|
Lewis Structure for Oxygen Difluoride SF4: Sulfur Tetrafluoride - Central Connecticut State University. Sulfur Difluoride Lewis Structure - Ask.com What Is Sulfur Difluoride Lewis Structure of Sulfur Dioxide Sulfur Monoxide Lewis Structure Sulfur Trifluoride Lewis Structure What does the Lewis dot structure look like for sulfur dioxide The gas sulfur difluoride is an unstable, reactive compound. Sulfur Tetrafluoride Lewis Structure Sulfur Difluoride Lewis Dot StructureSF4: Sulfur Tetrafluoride - Central Connecticut State University. Sulfur difluoride lewis structure Lewis Structure for Sulfur dichloride - College of DuPage - Home Lewis Structure: Sulfur Dioxide: SO 2: Lewis Structure The lewis structure of SF2 contains 16 nonbonding electrons and 4 bonding electrons. Lewis Structure: Xenon Difluoride: XeF 2: Lewis. SF2 Lewis structure is made up of two atoms, sulfur, and fluorine, the sulfur is in the central position and fluorine atoms are on either side of it. There are a total of 20 valence electrons: 6 by sulfur, click for more FAQs. Similar to his longtime best friend and rival Ryu, Kens goal is to test his power against many different fighters and strives to become stronger, but holds more restraint due to not wanting to jeopardize his family life. 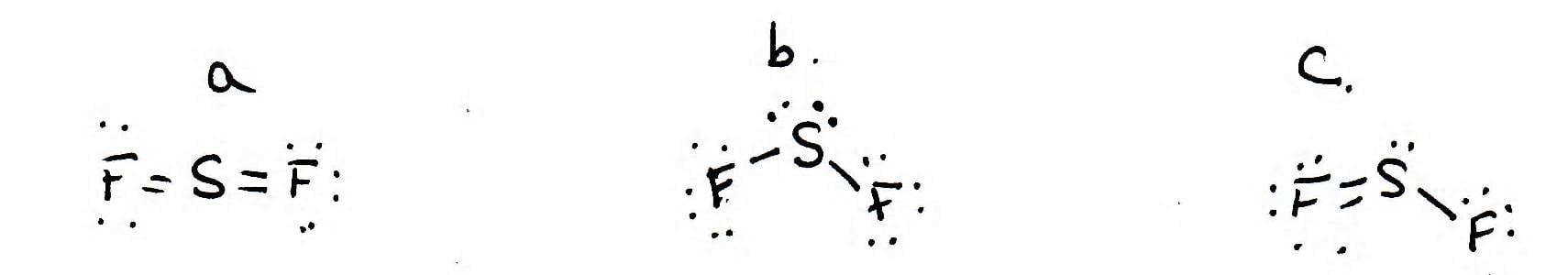
SF2 FAQ - What is the Lewis dot structure for SF2? - SF2 forms a bent compound. Ken Masters (, Ken Masutzu) is the deuteragonist of the Street Fighter series, first appearing in the original Street Fighter. Lewis Structures - Purdue University College of Science Welcome Sulfur Difluoride Lewis Structure - Ask.com Find 32 questions and answers about Sulfur Difluoride Lewis Structure at Ask.com Read more. Top questions and answers about Sulfur Difluoride Lewis Structure. SF2 has a simple Lewis structure in which the Sulphur atom is in the centre forming single bonds with both the Fluorine atoms.There are two lone pairs of electrons on the Sulphur atom which makes the geometry of the molecule bent.The Sulphur atom has sp3 Hybridization, and the bond angle of F-S-F is 98 degrees.It is a polar molecule as there is. The gas sulfur difluoride is an unstable, reactive compound. i) Draw the Lewis structure of sulfur tetrafluoride The gas sulfur difluoride is an unstable, reactive compound that exists asmonomer. The S is in the same family as O, but is larger than O, so the molecular shape is bent and the lone pairs are farther from the. The only difference between these Lewis structures is the identity of the oxygen atom to which the double bond is. Two Lewis structures can be written for sulfur dioxide. Sulphur difluoride sulphide - WebElements Periodic Table of the. Your thinkquest does show them, but if you're just starting with. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
